What is SLMTA?
SLMTA is a PEPFAR flagship program for strengthening laboratory systems. It was launched in 2009 simultaneously with the SLIPTA program, which provides a stepwise approach to measuring progress towards accreditation. While SLIPTA measures the laboratory quality by conducting audits, SLMTA provides the how-to with training and mentoring. These 2 programs complement each other and together they provide the tools and processes needed to turn the aspirations of lab accreditation into reality. There is no doubt that the laboratory landscape has been transformed in the past 15 years.
SLMTA is not just one training curriculum, but a series of learning tools created overtime in response to the evolving laboratory needs.
Click to learn about the evolution of the SLMTA program & course offerings
Click to learn how SLMTA aided the fight against the COVID-19 pandemic
Accomplishment To Date
To date, SLMTA has been implemented in 1643 laboratories in 56 countries in Africa, Asia, Latin America, the Caribbean, and even the Oceania. It has helped 432 laboratories in 31 countries attain accreditation to international standards.
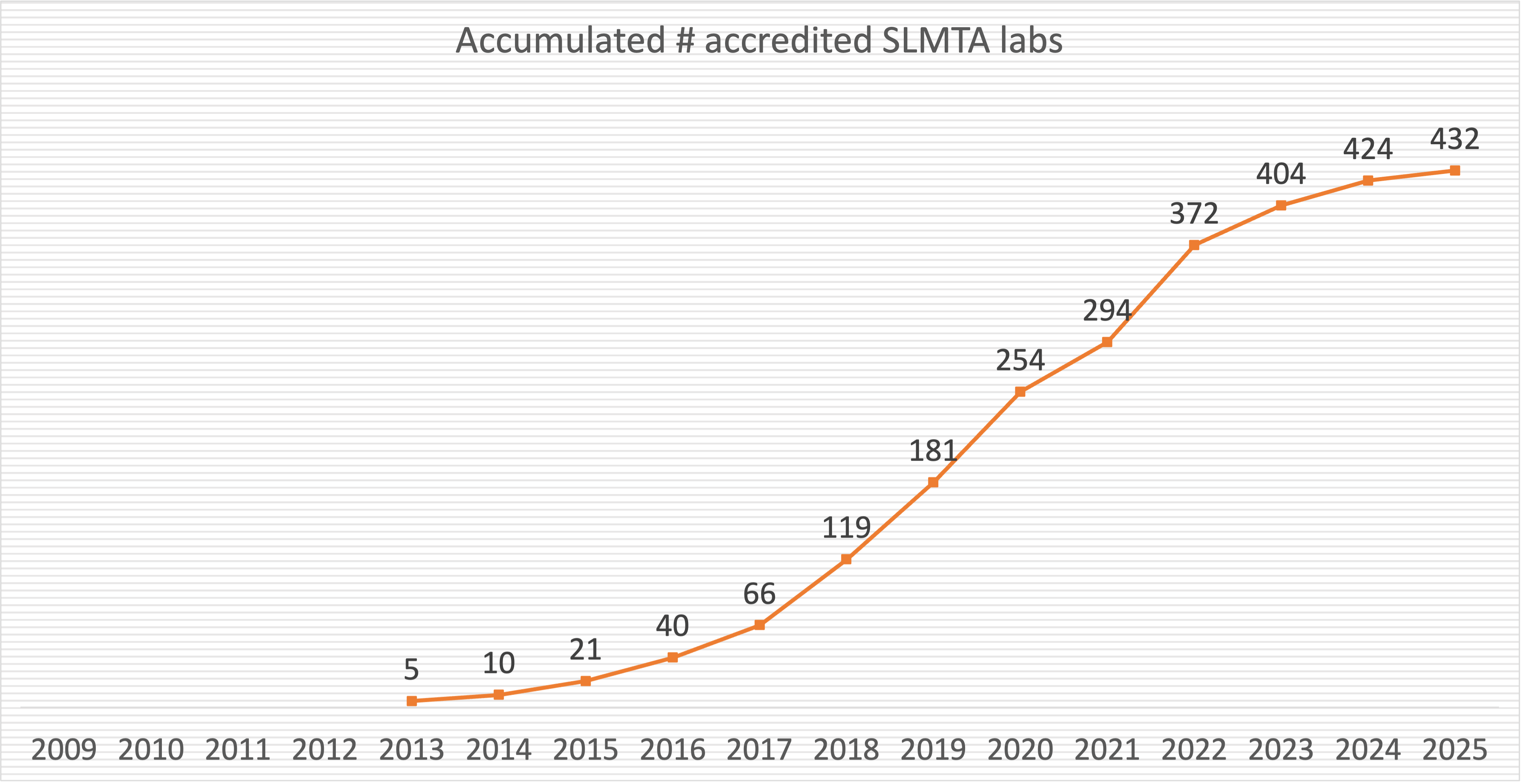
See all Accredited Labs
What do we want to achieve ?
- Vision
- Mission
- Goals
Better patient outcomes through sustainable quality laboratory systems and universal access to accurate, reliable, and timely laboratory results for correct diagnosis worldwide.
To transform the laboratory system by strengthening quality management systems aligned to international standards using a continuous quality improvement approach, and by fostering a quality culture based on excellence of services.
To implement the SLMTA/ SLIPTA* program and set laboratories on their path to accreditation as a measure to assure quality diagnosis, disease surveillance and outbreak response.
*SLIPTA - Stepwise Laboratory Quality Improvement Process Towards Accreditation
My SLMTA Story





















Listen to the SLMTA Songs





























 Do you have a SLMTA hero you want to nominate? Download the form
Do you have a SLMTA hero you want to nominate? Download the form


















